TREATMENT OF INDUSTRIAL WATER WITH FLOCCULATION THROUGH ELECTROCOAGULATION
The theory
The flocculation consists of a physical-chemical process that leads to the formation of a colloidal system, in which the solid phase tends to separate forming some flakes in suspension.
The process is based on adsorption phenomena where pH, temperature and ionic strength are environmental factors that strongly influence the flocculation, but also simply the formation of aggregates, or the flakes in suspension through the precipitation of dehydrated elements.
At plant level, flocculation is exploited in water purification treatment of both raw and wastewater, especially when you want to remove colloidal particles hardly settleable or filterable.
In order for the flocculation occurs, you must first destabilize the charges of the colloids.
This is achieved by making use of coagulants (the term is often used interchangeably flocculants).
These are compounds of various origin that exert their action in the range of pH values well defined.
A sufficiently high temperature, an increase of ionic strength and a strong agitation are factors that promote clotting.
The Basic Principle
The electrocoagulation is a method already known for a long time for the treatment of industrial waste water.
In its essential lines consists of an electrolytic cell where are positioned some plates of aluminum electrode, that by the application of a suitable electric current dissolve slowly entering into solution with the water to be treated (anodic oxidation).
These metal ions, in the form of hydrates, absorb the pollutant material creating floccules, while the cathode is the formation of hydrogen gas (cathodic reduction), which gives the possibility to the formats floccules to remain afloat.
Our solution involves the construction of an electrochemical cell where the continuous current passes through the plates of a particular aluminum alloy suitably sized to obtain the maximum yield.
In this process, the following reactions occur:
Anode: Al0 = Al3+ + 3e
Cathode: H0+2e = 2 OH + H2
The effect of the electric field is such as to cause the crushing also of large organic molecules of the compounds present in the water to be treated (for example ethoxylated or non-ionic surfactants) in fractions to lower molecular weight, so demolishing the active ingredient and more in general determining the reduction of soluble C.O.D.
The combined effect of the three actions above described:
- demolition of macromolecules,
- formation of coagulants centers such as hydroxides of metals already present in the solution or provided by solubilization of the electrodes,
- development of micro-bubbles of gas that rising on the surface drag emulsions, colloids and flakes in training
clearly has a synergistic action on the flocculation in place, thus increasing substantially the performance of the system.
The unwanted contaminants are subsequently removed by sedimentation and filtration.
The innovation
Compared to the water treatment with aluminum sulfate or aluminium poly-chloride, the electrochemical generation of aluminum has several advantages.
The aluminum is introduced without the corresponding sulfate ions (or chloride).
Furthermore, there is no need to supply alkalinity to give a reaction.
Eliminating competing anions through the use of a source of high purity aluminum, you get the metal residues considerably lower and, consequently, less sludge is produced (50 ÷ 70% less).
The adjustment of the aluminum ions dose in water can be performed easily with the knob for the control of the current.
The amount of dissolved aluminum depends on the quantity of electricity that passes through the water, and may be derived from Faraday's law:
M = 1/96500 · A / V · l · t
where:
M = mass of dissolved aluminum
A = atomic mass of aluminum (27)
V = valence of aluminum (Al +++=3)
I = current in ampere
t = time in seconds
Besides both the anode and the cathode chemically dissolve in water:
Al0 + 3H2 = Al (OH)3 + 1.5H2
The nascent Al + + + reacts with water to form insoluble hydroxides and then the process of destabilization of the colloid is similar to that obtained with traditional metal salts.
For the full treatment of wastewater, the electro-cell is followed by the usual separation processes.
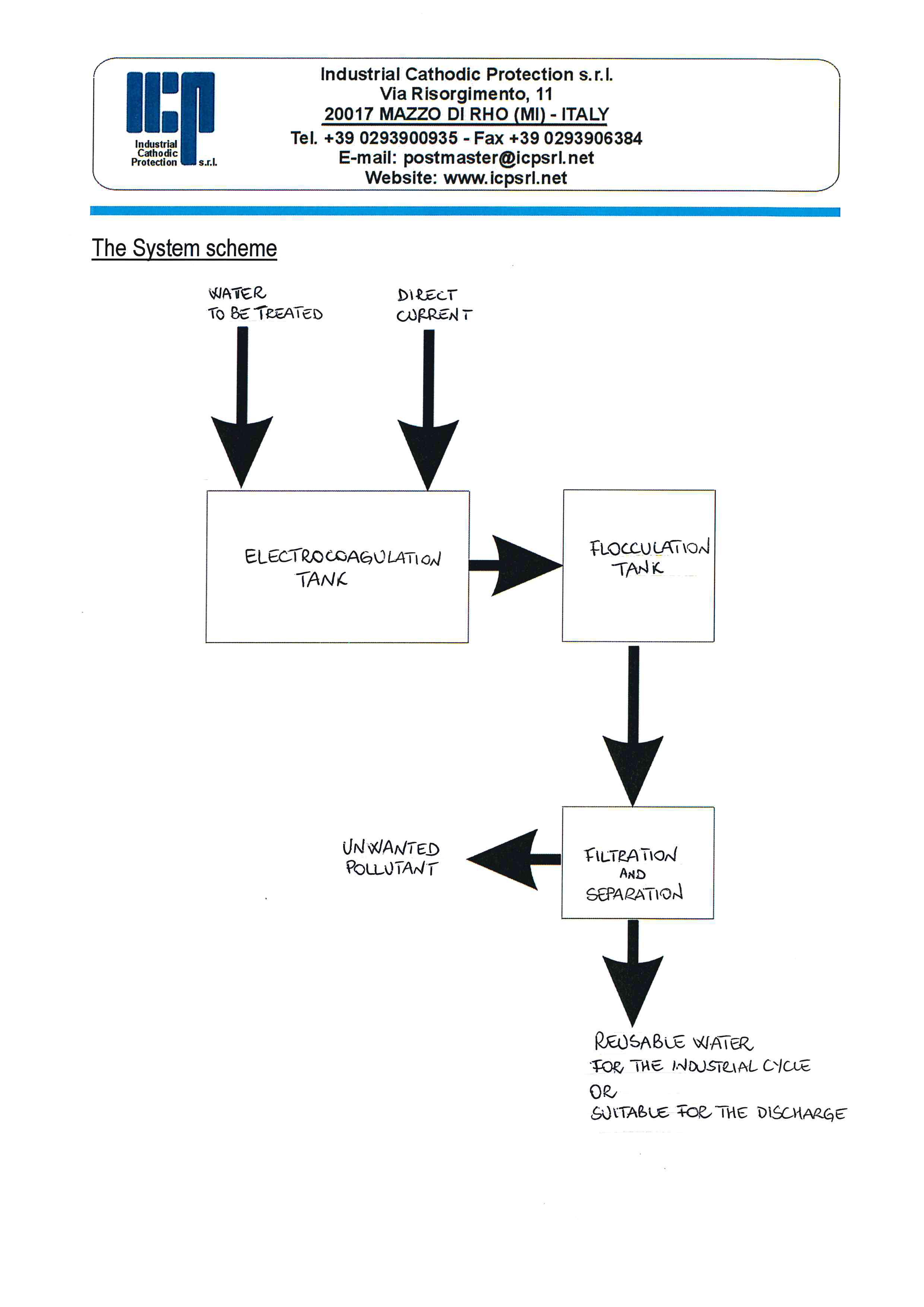
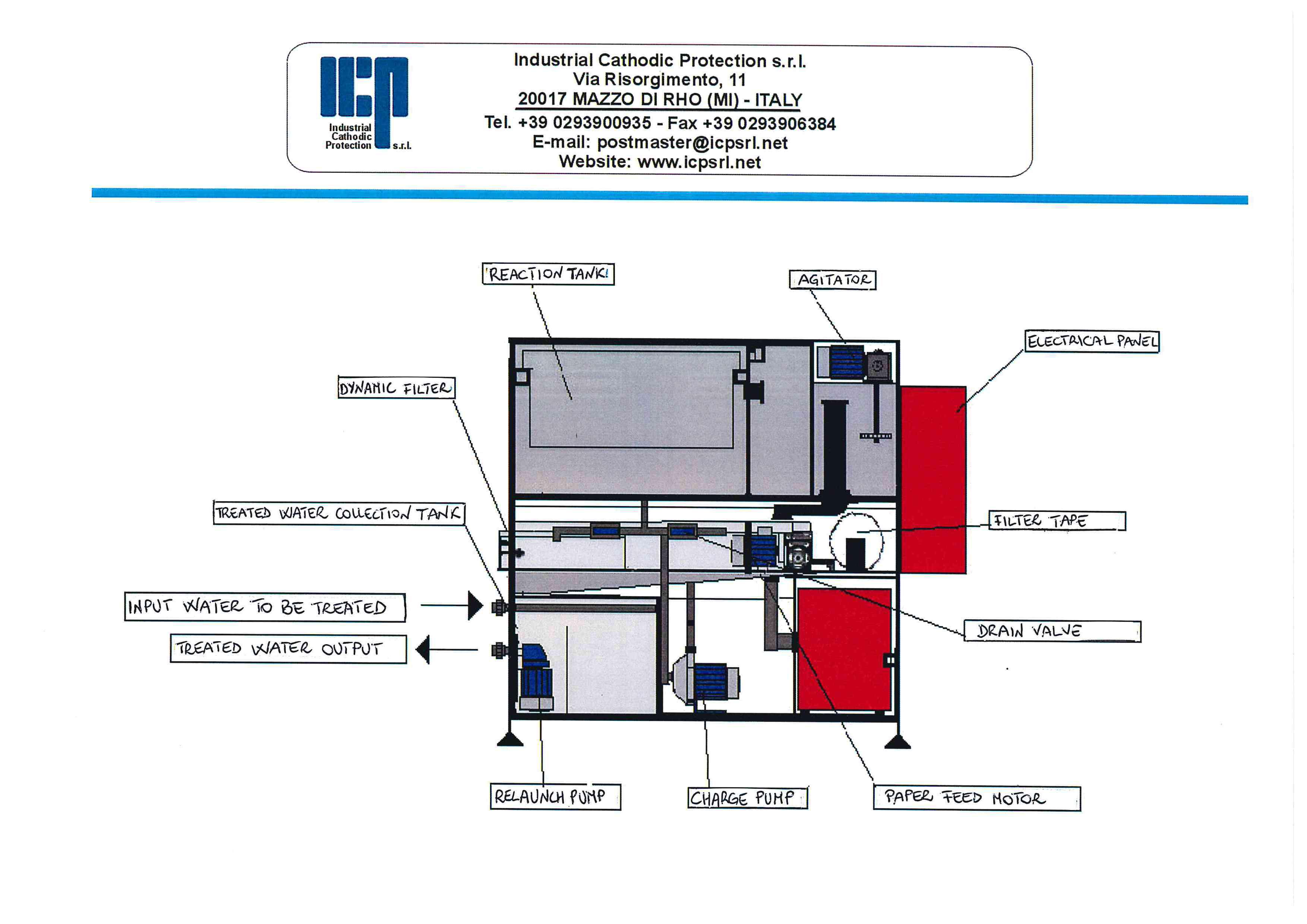
Electrocoagulation tank
In the tank are placed electrodes that release aluminum hydroxide under the effect of the current flowing.
The size and design are calculated on the basis of required flow.
The electrodes are connected by aluminium rods placed on the edge of the tank.
The connection allows to send electric current to the individual electrode, in this way it is created the single cell.
The basic principle is the following:
The system will absorb a current proportional to the conductivity of the waste water; according to the Faraway’s law the absorbed amps will dissolve a proportional amount of aluminum.
Flocculation tank
Overflowing, the treated wastewater will pass to the flocculation tank where a polymer is added to allow aggregation groups of larger size.
Filtering
Overflowing, a roll of filter paper will receive the water / groups in suspension and will allow the filtering.
When the level of the suspension will come to touch the upper level, it means that the filtering speed becomes too low, thus, the paper will advance automatically in order to increase the filtering speed.
Paper and sludge will be collected together in a plastic container.
The filtered and treated water is collected in a temporary tank where a drain pump sends the filtered water to a storage tank for the re-use or discharge.
Control Panel
A control panel manages all the automated process of checking every single detail and blocking the process in the event of any anomalies
Technological benefits
This system is intended for the treatment of highly polluted waste water, especially when water re-use is required.
The main advantages of this method are:
- LOWER investment respect to the competing technology (evaporators or chemical / physical)
- NO change of water parameters (pH and conductivity do not change)
- INCONSIDERABLE use of chemical products
- SMALL production of resulting sludge
- LOW maintenance costs
- LOW consumption of electricity
- PRODUCTS absolutely not dangerous
- Abatement PERFORMANCE consistently superior to the traditional chemical-physical treatment
- SIZE of the plant extremely contained
Characteristics of our plants
Our main machine ELECO1000, currently used for the treatment of flexography waters, has the following characteristics:
- Dimensions: 2500x1700x2800 mm.
- Maximum current applied: 12 V / 240 A
- Electrodes: 34 = 13.6 square meters
- Production capacity: 750-1000 liters / hour (depending on the quality of waste water)
- Sludge formation: 100 g / hour
- Average humidity sludge: 75%
- Polymer consumption: 20 gr / hour
- Power maximum consumption of the electro-cell: 2.88 Kw / hour
It is also available a reduced version ELECO400 with production capacity: 200-400 liters / hour.
The versatility of the electrocoagulation system allows us to design and build machines at the specific request of the customer, depending on the type and flow of waste water to be treated.